MF.Asia: Your Comprehensive Guide on Medical Gowns

In the realm of healthcare, the use of personal protective equipment (PPE) is essential to ensure the safety and well-being of healthcare workers and patients. Among the diverse array of PPE, medical gowns hold a significant position. These gowns play a vital role in various healthcare settings, ranging from surgical procedures to routine patient care, and are particularly crucial in the face of contagious diseases.
Medical gowns provide a comprehensive shield, offering protection against infectious materials and minimizing the risk of transmission. However, to make well-informed decisions and maintain effective infection control protocols, it is imperative to have a thorough understanding of the different types of medical gowns, as well as the applicable standards and regulations governing their use.
In this article, we will also uncover the key factors to consider when choosing the right medical gowns for your clinic or hospital. Factors such as material selection, level of protection, comfort, and ease of use will be highlighted to aid you in making informed decisions that align with the specific needs of your healthcare setting.
By the end of this guide, you will be equipped with the knowledge necessary to confidently select and procure medical gowns.
Understanding the Role of Medical Gowns in Infection Prevention
Medical gowns are the unsung heroes that ensure safety and confidence in the medical field used in a variety of healthcare settings, including hospitals, clinics, and laboratories.
It acts as a sturdy barrier, shielding the wearer's skin and clothing from exposure to blood, body fluids, and other potentially infectious materials (OPIM). Their primary purpose is to mitigate the risk of infection transmission during surgical procedures and the care of patients with contagious diseases. They are a vital component of the comprehensive infection-control strategy employed in healthcare facilities worldwide, preventing direct contact and reducing the risk of transmission. Additionally, medical gowns prevent the wearer from inadvertently transferring harmful microorganisms to vulnerable patients, particularly those with compromised immune systems.
Types of Medical Gowns
Medical gowns used in healthcare settings go by various names, including coveralls. Each type serves a specific purpose, tailored to the demands of different medical scenarios. Whether it's a high-risk surgical procedure or routine patient care, medical gowns provide a broad spectrum of protection, instilling confidence in healthcare professionals and ensuring patient safety. Generally, medical gowns can be broadly classified into three categories including:
Isolation Gowns:
Isolation gowns, also known as non-surgical isolation gowns, are intended for low or minimal risk situations designed to cover most of the body, providing a high level of protection against the transmission of microorganisms and OPIM. The FDA classifies isolation gowns as Class I medical devices, which are exempted from premarket 510(k) notification requirements.These are not worn during surgical procedures, invasive procedures, or in situations with a medium to high risk of contamination.
Furthermore, non-surgical non-isolation gowns are intended for use by healthcare personnel, non-surgical, and non-patient isolation situations. They are not used in a sterile field, such as during surgery, which can be reused if laundered using enzymatic detergent or according to the hospital's standard operating procedures.
Surgical Isolation Gowns:
Within the category of isolation gowns, there exists a specialized sub-category referred to as "surgical isolation gowns," which are specifically designed to offer a higher level of barrier protection in situations involving moderate to high risk.
Similar to surgical gowns, surgical isolation gowns fall under the FDA's Class II medical devices, requiring a 510(k) premarket notification. However, surgical isolation gowns require larger critical zones to provide enhanced coverage and protection. The critical zones as shown in the image below must meet the highest liquid barrier protection level specified for both surgical and non-surgical isolation gowns. The liquid barrier protection of all seams must be on par with the rest of the gown. Furthermore, the fabric of the surgical isolation gown should cover the body adequately according to the intended use.
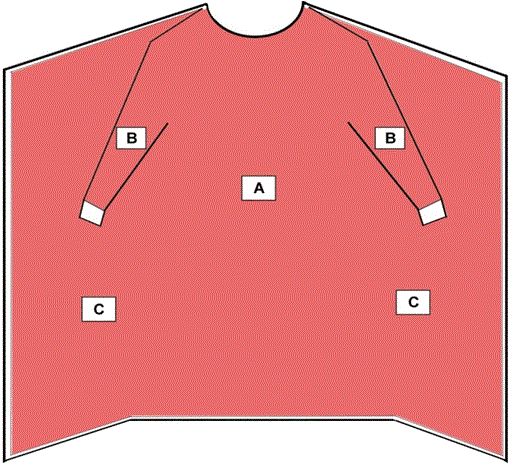
Image extracted from the FDA Medical Gowns
Critical zones for surgical and non-surgical isolation gowns in areas A,B, and C including seams but excluding cuffs, hems, and bindings, designed with larger critical zones than surgical gowns. Barrier performance of at least Level 1 is required.
Surgical Gowns:
Surgical gowns are specifically designed for use during surgical procedures. They serve a dual purpose: a) protecting the surgical staff from potential contamination by the patient's body fluids and tissues, and b) preventing the patient from being exposed to the surgical staff's skin cells, hair, and microbiota, which could potentially cause surgical site infections.
Surgical gowns are classified as Class II medical devices by the FDA, indicating they are considered intermediate risk. They must be cleared through the premarket notification process to demonstrate that the device is safe and effective before it can be marketed.
National standards have defined critical zones of protection for surgical gowns, as illustrated below. These critical zones encompass the front of the body from the top of the shoulders to the knees, as well as the arms from the wrist cuff to above the elbow. Surgical gowns must be sterile and appropriately labeled.
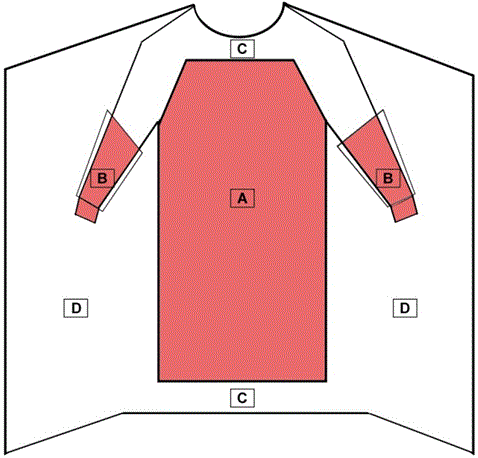
Image extracted from the FDA Medical Gowns
Critical zones for surgical gowns in areas A, B, and C is required to have a barrier performance of at least level 1. The back of the surgical gown (area D) may be non-protective.
Disposable Coveralls:

Image extracted from the CDC
(White coveralls vs yellow isolation gowns)
Coveralls are full-body suits that provide full coverage from the neck down, including the torso, arms, and legs. They are often used in situations where there is a higher risk of exposure to hazardous chemicals or biological agents. Coveralls usually have integrated hoods, boots, and elasticized cuffs for complete coverage against various hazards such as chemicals, biological agents, particulates, and liquids.
The choice of material depends on the specific requirements of the task or environment. Some common materials used for coveralls include polypropylene (general-purpose), microporous film laminate, and/or flame-resistant materials.
Disposable coveralls are designed for single-use and are typically made from non-woven materials. To ensure maximum protection, it's essential to use and care for coveralls correctly. Here are a few key points to keep in mind:
- Proper Sizing: Coveralls should be selected in the correct size to provide a comfortable fit and ensure full coverage.
- Donning and Doffing: Follow CDC PPE sequence for putting on (donning) and removing (doffing) coveralls to minimize the risk of contamination.
- Disposal: Disposable coveralls should be properly disposed of according to local regulations and guidelines. This helps prevent the spread of contaminants.
For more information on the national standards and guidelines provided by the Ministry of Health, Singapore, in different care settings, please visit the “National Infection and Prevention Control Guidelines and Standards” page and download the relevant guidelines.
Looking to purchase coveralls? Visit your local medical supply store today at Mf.Asia Disposable Non-Sterile Coverall.
Standards and Regulations

Medical gowns are subject to various standards and regulations to ensure their safety and effectiveness. These standards are set by several organizations, including the FDA, the Occupational Safety and Health Administration (OSHA), and the Centers for Disease Control and Prevention (CDC). The FDA regulates the manufacture and sale of PPE, while OSHA enforces the proper use of PPE in healthcare facilities. The CDC provides guidelines for the use of PPE, including isolation gowns, in healthcare settings.
4 Levels of American National Standards Institute/Association for the Advancement of Medical Instrumentation (ANSI/AAMI) PB70 Standard
When selecting gowns, it is important to consider the intended use and the level of protection required. The FDA recognizes the ANSI/AAMI PB70 standard, which classifies protective apparel and drapes based on their liquid barrier performance. This standard includes four levels of barrier performance, with level 1 being the lowest and level 4 being the highest test methods to determine compliance. These tests evaluate the gown's resistance to water, blood, and other fluids, as well as its seam strength and tear resistance. The classification system includes the following levels:
Level 1: Minimal risk
Suitable for basic care, standard isolation, visitor cover gowns, or in a standard medical unit.
Level 2: Low risk
Appropriate for activities like blood draw, suturing, in the ICU, or in a pathology lab.
Level 3: Moderate risk
Recommended for procedures such as arterial blood draw, IV line insertion, in the ER, or for trauma cases.
Level 4: High risk
Designed for long, fluid intense procedures, surgeries, situations requiring pathogen resistance, or suspected infectious diseases (non-airborne).
By understanding these risk levels and referring to the product labeling, you can make informed decisions when choosing the most suitable protective gowns for specific tasks or situations.
European Norms (EN) 13795 and 14126 Standards
When classifying protective clothing, two European standards play a significant role. EN 13795 is an essential standard that outlines the requirements for surgical gowns, drapes, and clean air suits utilized in operating rooms. It offers comprehensive guidelines for the design, performance, and labeling of these products, encompassing aspects such as liquid resistance, microbial barrier performance, linting, and cleanliness.
On the other hand, EN 14126 focuses specifically on protective clothing against infective agents. It sets rigorous requirements for clothing materials to act as effective barriers against biological hazards like viruses, bacteria, and other microorganisms. This standard evaluates the clothing's resistathis website from the CDC.nce to blood and body fluid penetration while assessing its ability to prevent the transmission of pathogens. These European standards ensure that protective clothing meets the necessary criteria to provide appropriate levels of protection in healthcare and other high-risk environments.
The National Fire Protection Association (NFPA) 1999 Standard
The NFPA 1999 developed this standard specifically for protective clothing used by emergency medical services (EMS) personnel. NFPA 1999 outlines the requirements for EMS garments, including coveralls, to protect against biological hazards and other threats. It covers criteria such as barrier performance, durability, and flame resistance.
Typically, when it comes to classifying protective clothing, gowns are assessed using ANSI/AAMI PB70 and EN 13795 standards, while coveralls are evaluated using EN 14126 and NFPA 1999.
For a comprehensive understanding, please refer to this piece of research journal for additional information on laboratory studies, standards, and regulations pertaining to medical gowns and the differences in the test standard methods according to the CDC. For more PPE FAQs, please visit this page from the CDC.
Choosing the Right Medical Gowns

In healthcare settings, selecting the appropriate PPE is paramount to ensure the safety of both healthcare providers and patients. When it comes to medical gowns, making the right choice is crucial in providing effective barrier protection. Several factors should be taken into account such as:
- Biocompatibility Assessment: Medical gowns are considered surface-contacting devices, making it essential to evaluate their biocompatibility. The FDA recommends assessing cytotoxicity, sensitization, and irritation for these gowns. By adhering to these guidelines, healthcare facilities can ensure that the gowns are safe for use. For more detailed information, the FDA's Biocompatibility Assessment Resource Center is a valuable resource.
- Check for FDA Clearance and Product Codes: To identify FDA-cleared medical gowns, you can refer to the 510(k) Premarket Notification database. Look for product codes such as FYA, FYB, FYC, and QPC to find approved gowns. This step ensures that the gowns meet the necessary standards and regulations set by the FDA.
- Consider the 4E model (Evaluate, Engage, Educate, Execute) guided by the CDC: click here for more information Guidance for the Selection and use of PPE in Healthcare Settings.
- Type of Procedures: If your clinic performs surgeries or procedures with a high risk of fluid splash or exposure to infectious materials, surgical gowns with a high barrier performance (Level 3 or 4) would be appropriate. For standard patient care and minor procedures with low fluid exposure, isolation gowns or surgical gowns with lower barrier performance (Level 1 or 2) may suffice.
- Potential Exposure: Assess the potential exposure to blood, body fluids, and other infectious materials. If the risk of exposure is high, it is essential to choose a gown with a higher barrier performance to provide increased protection.
- Comfort and Flexibility: Consider the comfort and flexibility of the gowns, particularly during procedures that require movement and dexterity. Breathable gowns that allow air and moisture vapor to pass through can enhance comfort, especially during longer procedures.
- Checking Expiration Dates: Always check the product labeling for the manufacturer-designated shelf life of the gowns. Expired gowns should not be used for procedures requiring barrier protection but can be utilized for training and demonstration purposes where barrier protection is not necessary.
- Resistance to Fluids: A crucial characteristic of medical gowns is their ability to repel fluids, preventing penetration and minimizing the risk of contamination.
- Breathability: Gowns should allow airflow to reduce heat buildup and maintain comfort during prolonged use.
- Tear Resistance: Gowns should be durable enough to withstand the rigors of medical procedures without compromising their integrity.
- Size and Fit: Proper sizing ensures that the gown covers the healthcare professional adequately, reducing the risk of exposure.
Stay informed, follow the recommended guidelines, and make informed choices when it comes to medical gowns, as they play a crucial role in infection control.
Discover a wide range of patient and healthcare worker gowns in MF.Asia's catalog. Browse through our selection and find the perfect gowns to ensure safety and protection in healthcare settings.
Conclusion
As the backbone of infection control, medical gowns continue to evolve, adapting to the ever-changing landscape of healthcare. Stay informed, stay protected, and let medical gowns be your armor in the fight against infectious diseases. Understanding the types of gowns, their uses, and the standards and regulations governing their use is crucial for making the right choice for your clinic and/or hospital environment.
Experience the difference in safety and well-being with MF.Asia. Visit our website today to explore a wide range of high-quality PPEs. Your safety is our top priority, and we are dedicated to providing you with reliable and effective solutions. Thank you for taking the time to read this guide. If you have any further inquiries or need more information, please don't hesitate to contact us. Our knowledgeable team is here to assist you every step of the way.
Together, let's prioritize safety and create a healthier future. Visit MF.Asia now and discover the PPEs that will keep you protected.
View our full catalogue here.